Introduction
Age-related macular disease (AMD) is a disease that causes irreversible damage to the retina. AMD is categorized into two types: dry AMD and wet AMD. AMD accounts for 6–9% of blindness globally and stands as the leading cause of vision loss for over 50-year-old populations [1]. There are many factors that contribute to the progression of AMD, such as aging, smoking and obesity. Therefore, both dry and wet AMD are highly heterogeneous and can be affected by both genetic and environmental factors.
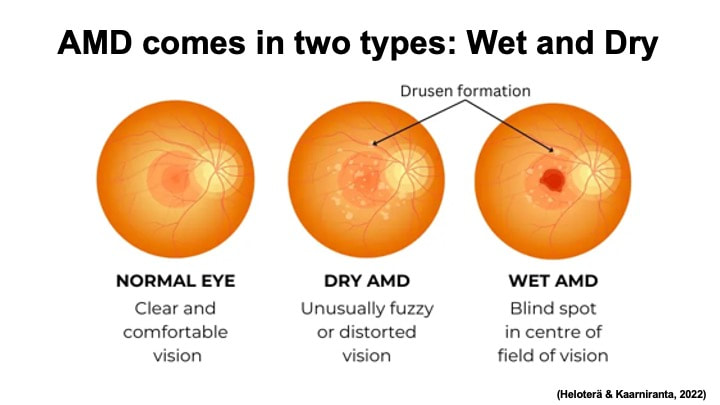
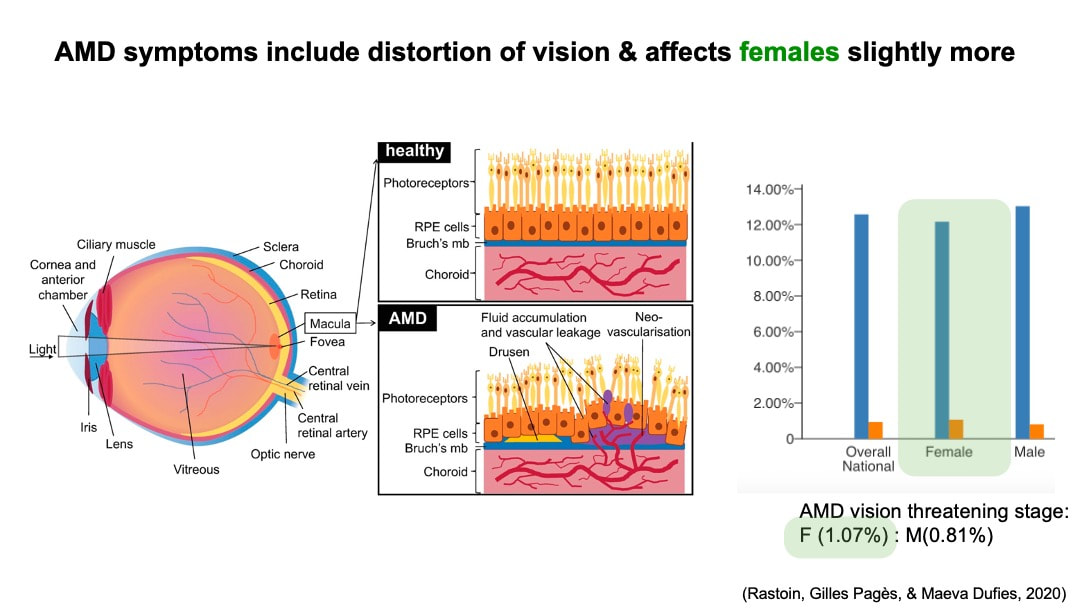
AMD comes in two different types: dry AMD or wet AMD, also known as neovascular or nAMD. Dry AMD is the most common form and is characterized by the formation of drusen between the retinal pigment epithelium (RPE) and Bruch’s membrane on the retina. Wet AMD is less common but can cause severe vision loss. As its other name neovascular AMD suggests, wet AMD is characterized by abnormal growth of blood vessel (angiogenesis) under the retina. However, it is worth noting that these two types of AMD are not independent from each other. Dry AMD usually appears as early stage of this disease and can progress to wet AMD in the later development.
AMD patients can use Amsler grid to keep track of the progression of AMD symptoms. To try it out, you can look at the center of the first grid picture below, covering one of your eyes. AMD patients usually see distortion of the lines.
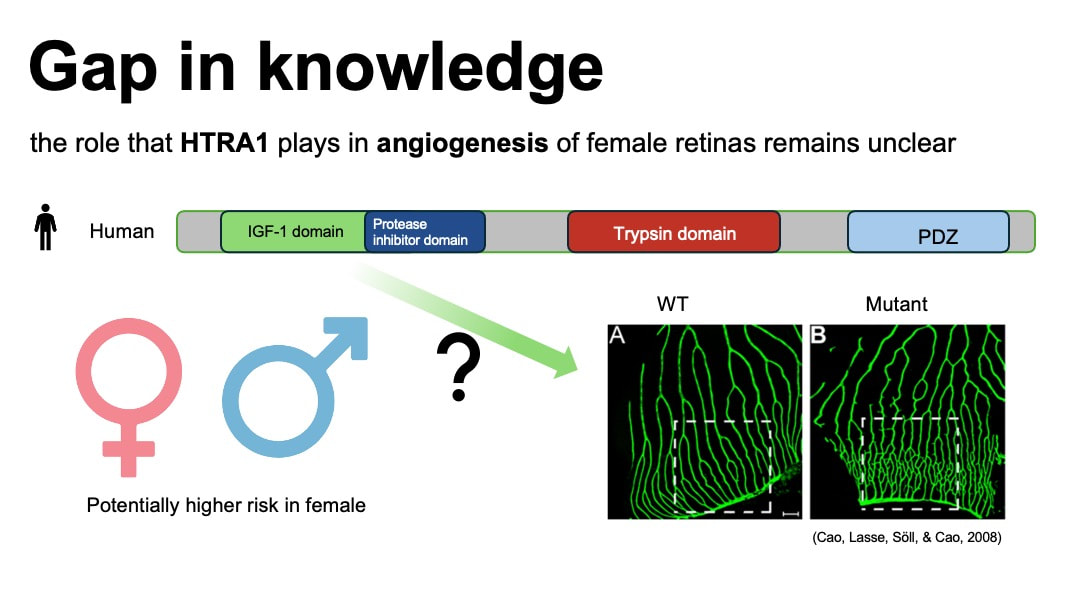
Anatomically, macular is part of the retina at the back of eyes and has a high concentration of photoreceptors. Therefore it plays an important role in the central vision. Although currently there is no data on the AMD gender prevalence difference in global population. Statistical analysis in gender difference of AMD in European population with an odd ratio of 1.24 [2] suggests a potential higher risk of wet AMD for female population. Based on the AMD national prevalence data collected in 2019 from The Centers for Disease Control and Prevention of US, there is slightly a higher prevalence for wet AMD in female.
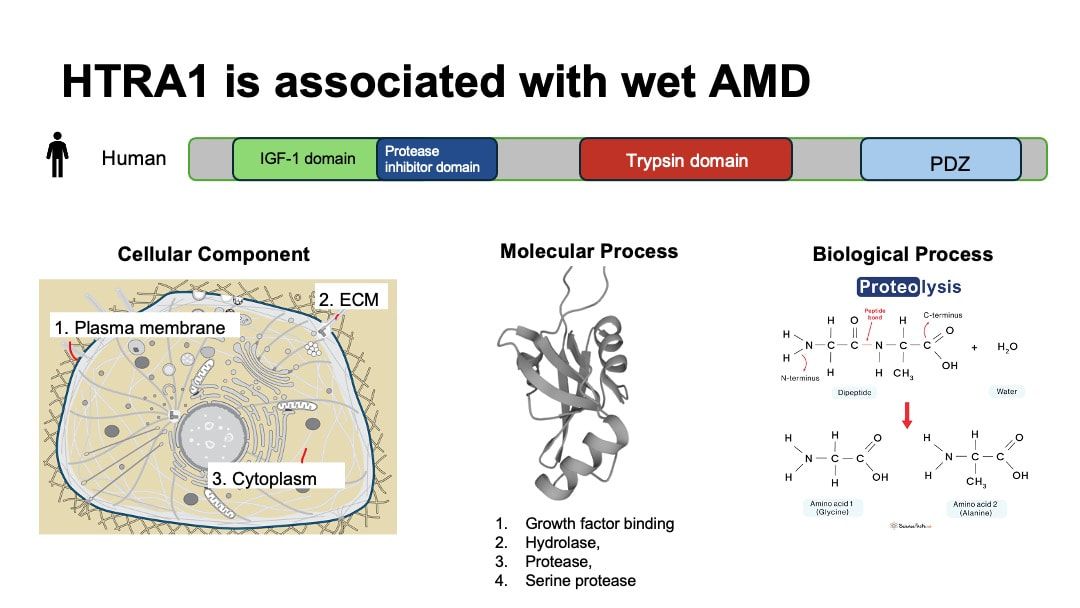
HTRA1 has emerged as a significant risk factor associated with wet AMD identified in genome wide association studies [3].HTRA1 gene encodes high-temperature requirement protein A1 (HtrA1).This protein is a serine protease secreted by a number of tissues including retinal pigment epithelium (RPE). HTRA1 is differentially expressed in female and male tissues. It is located in plasma membrane, ECM and cytosol. Its molecular function includes growth factor binding and serine-type endopeptidase activity. The main biological process that HTRA1 is involved is proteolysis.
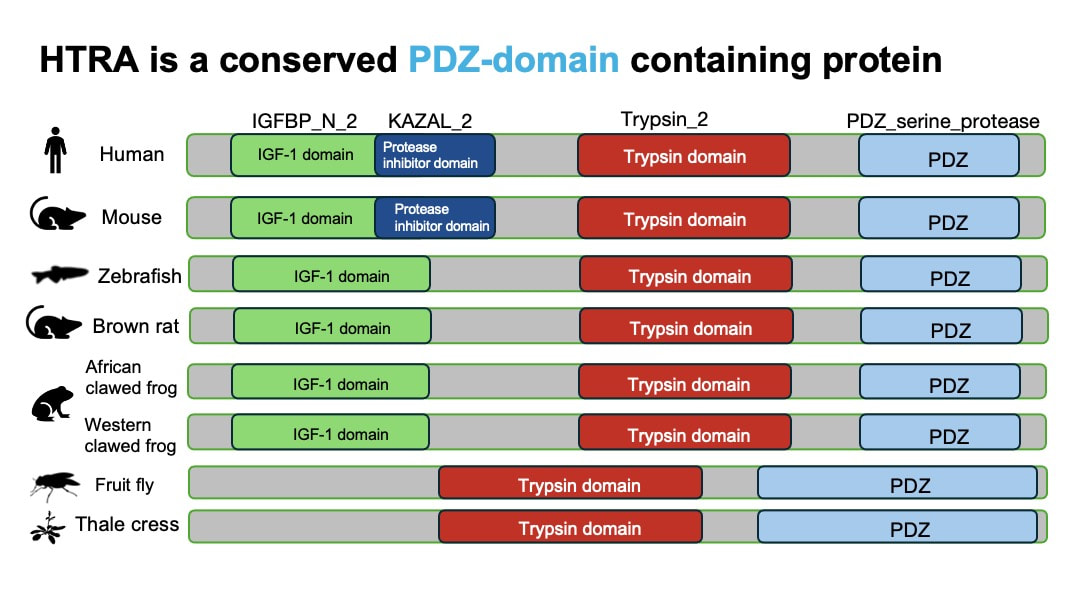
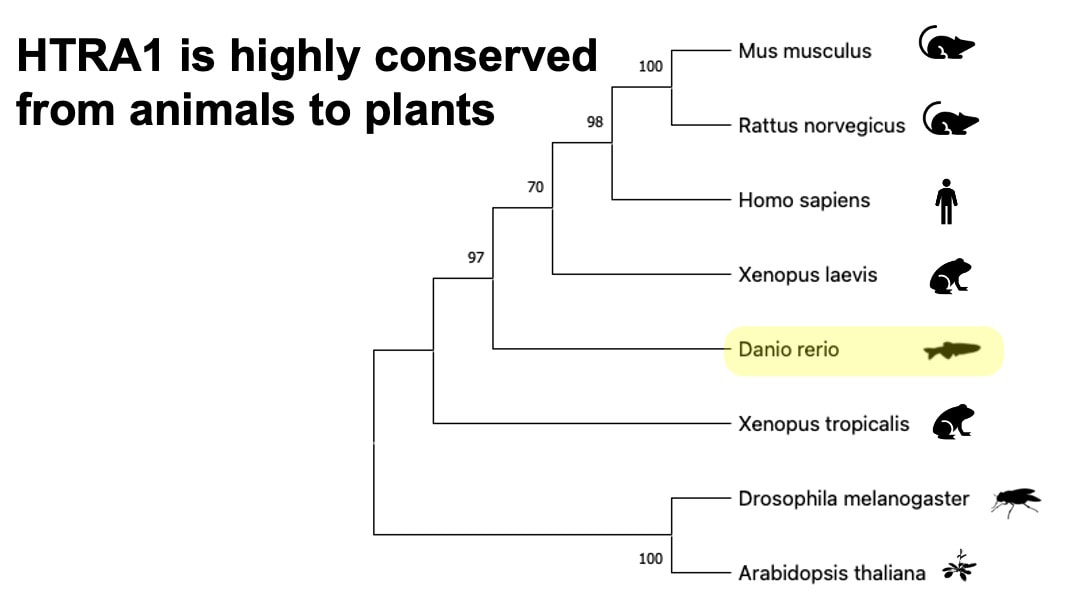
Based on the protein domain research from InterPro, HTRA1 is a conserved PDZ-domain containing protein. PDZ domain is associated with the function of serine protease. As is shown in the table below, organisms that are less phylogenetically distant from humans such as mouse, share more common protein domain with humans.
HTRA1 is also highly conserved from animals to plants.
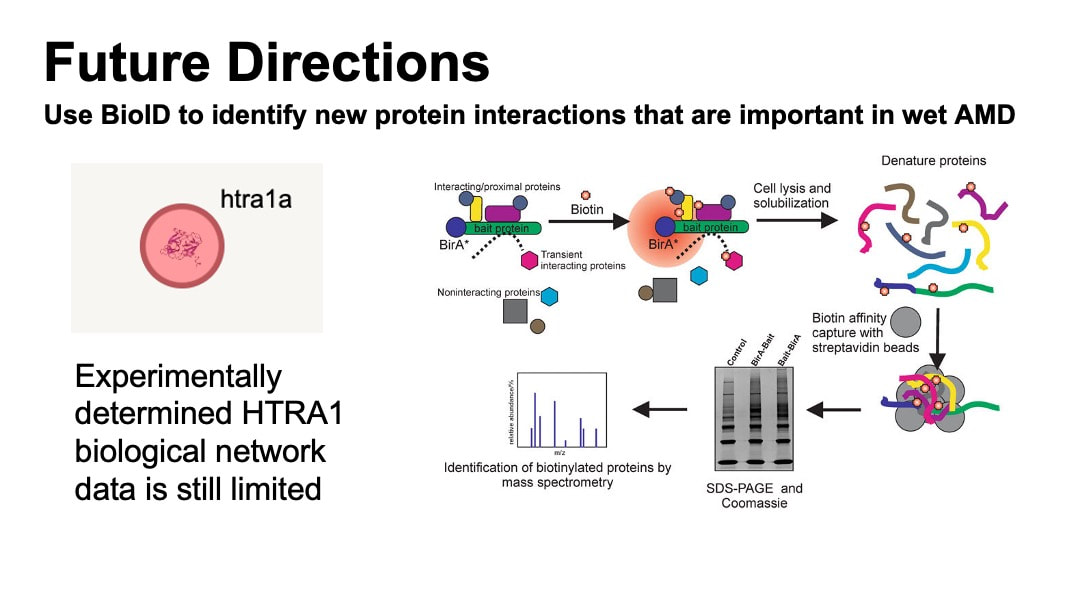
Protein interaction network data extracted from the STRING shows that HTRA1 interacts with protein involved in serine-type endopeptidase activity and complement binding. Regardlessly, the experimental data is still limited as there is no proteins associated with the GO terms of HTRA1 experimentally found to interact with HTRA1 in zebrafish.
There is no best animal model that recapitulates all aspects of AMD, as AMD is a complex disease involving various genetic, environmental factors. Each model organism has its advantage and disadvantage in reflecting different aspects of AMD such as oxidative stress, lipid metabolism and inflammation, etc. Therefore, it is important to have a combination of different animal models for the study of AMD.
In my study, I decided to use zebrafish as the model organism for the following reasons.
In my study, I decided to use zebrafish as the model organism for the following reasons.
- Zebrafish requires lower cost and high reproduction rates.
- Visualization of blood vessel in zebrafish is good for angiogenesis study.
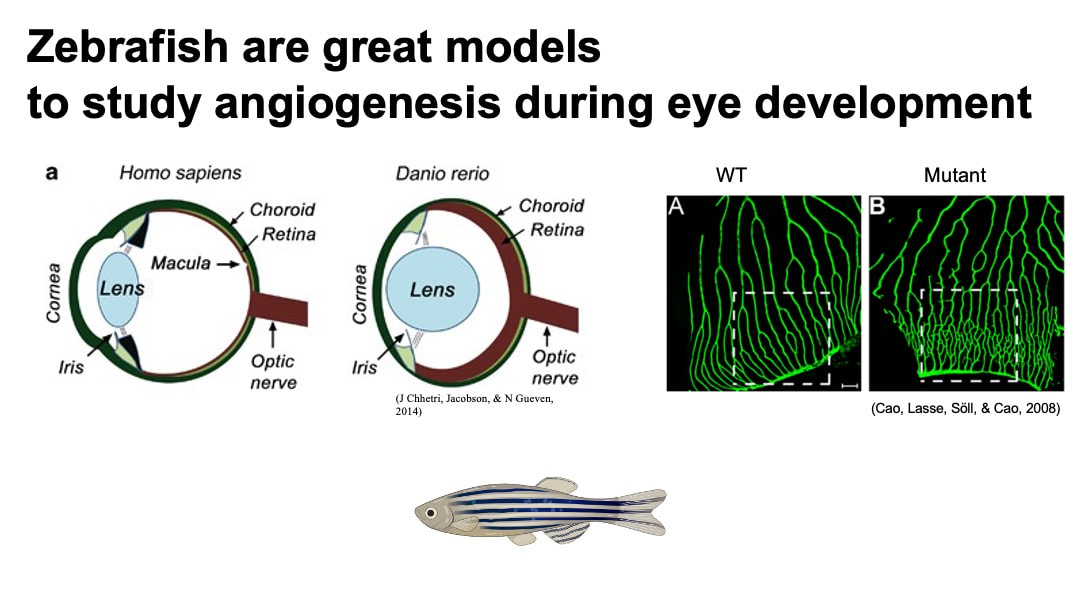
- Anatomically, zebrafish retina is cone-rich, which is similar to the human retina. This presents an advantage of zebrafish model for the study of AMD and other photoreceptor diseases over rodent models with rod-rich retina. Human macular is localized in retina and has highest concentration of cone photoreceptors across the retina. It is responsible for central, high-resolution, and color vision. [4] [6]
- Reliable model of neovascularization is able to be induced in zebrafish by putting them in hypoxia. [5]
- Around 70% of genes in the human genome have orthologs in the zebrafish genome. [5]
the role that HTRA1 plays in angiogenesis of female retinas remains unclear. Studying the molecular mechanisms of how HTRA1 functions in angiogenesis in the retina is important because it can define potential therapeutic targets for the wet AMD treatment, particularly in females.
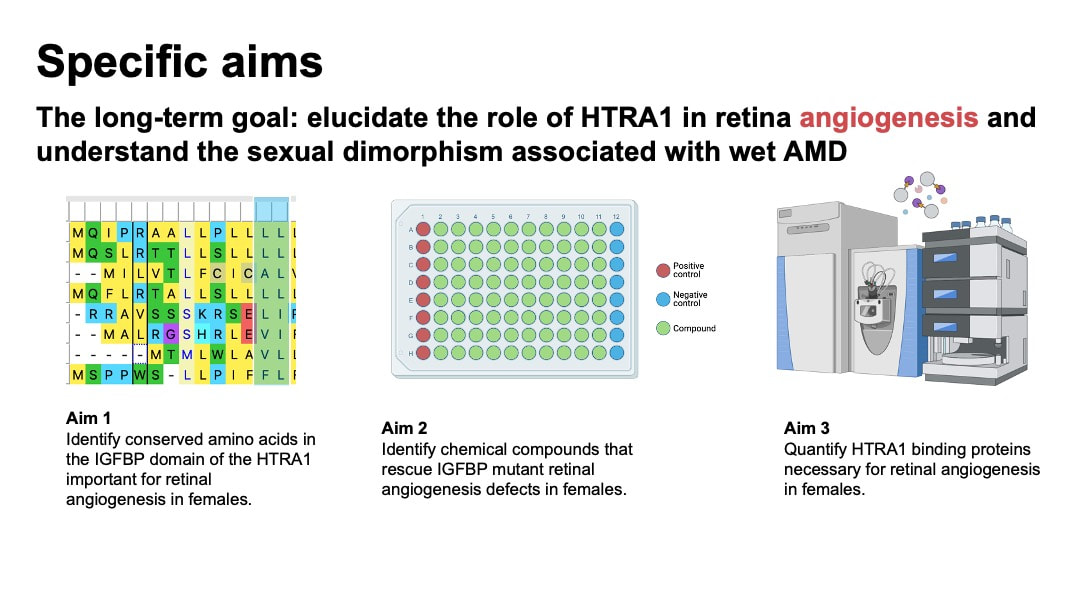
In my specific aims, The long-term goal is to elucidate the role of HTRA1 in retina angiogenesis and understand the sexual dimorphism associated with wet AMD. To achieve this, I will use zebrafish as model organisms, because they possess cone-rich retinas that resemble human retinas and blood vessel visualization is easy to assays. My objective is to characterize HTRA1’s function in retinal angiogenesis in zebrafish. I hypothesize that HTRA1 plays a critical role in angiogenesis of the retina of female by regulating the inflammatory and stress-related factors, and overexpression of HTRA1 upregulates inflammatory factors in retina, which eventually leads to angiogenesis.
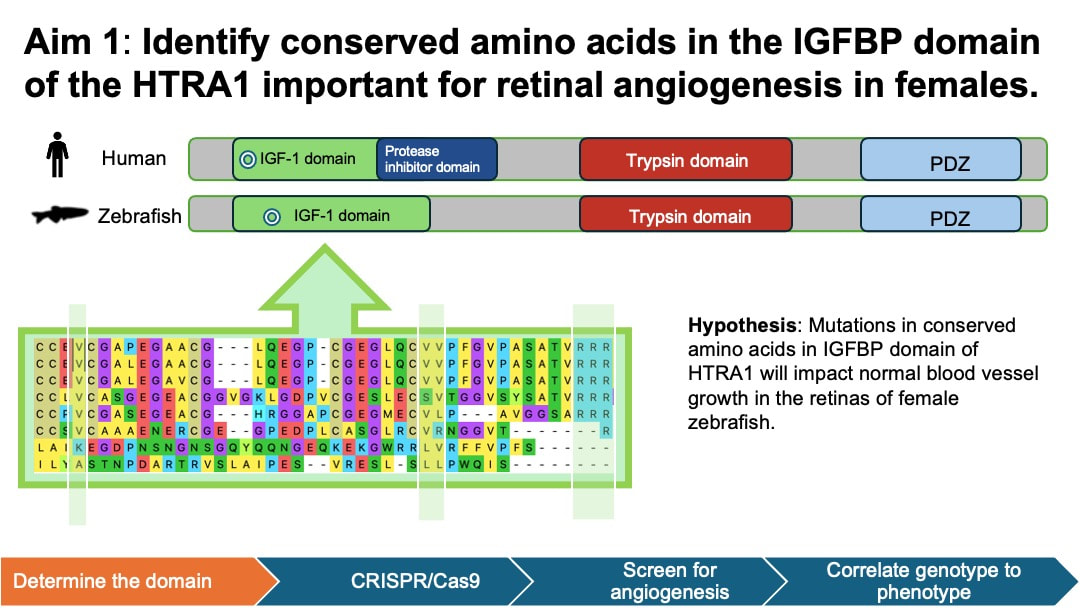
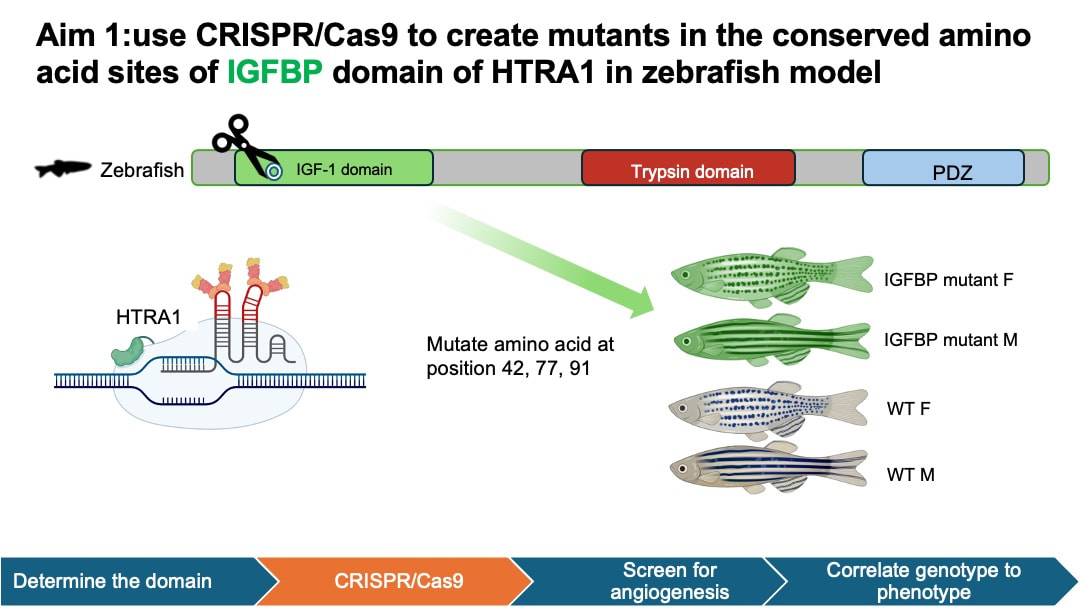
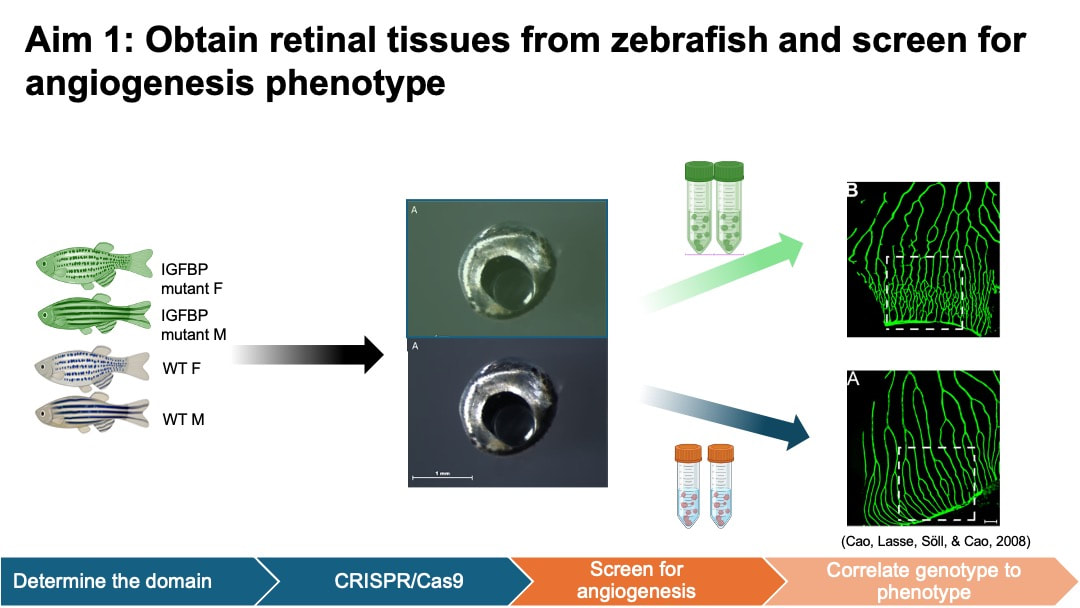
In my aim 1, I hypothesize that mutations in conserved amino acids in IGFBP domain of HTRA1 will impact normal blood vessel growth in the retinas of female zebrafish. I selected IGFBP as my target mutant because IGFBP domain is associated with growth factor binding and cell proliferation activity in the retina. This process is closely related to angiogenesis. To achieve this aim, I will determine the homology of HTRA1 gene domain using NCBI BLAST, followed by an examination of conserved amino acids using Multiple Sequence Alignment and ClustalOmega. Next, I will use CRISPR/Cas9 to create mutants in the conserved amino acid sites of IGFBP domain of HTRA1 in zebrafish model. Experimental zebrafish will be divided into 4 groups: wild-type (WT) male, WT female, mutant male, and mutant female. I will then screen the zebrafish to observe blood vessel development within their retinas.
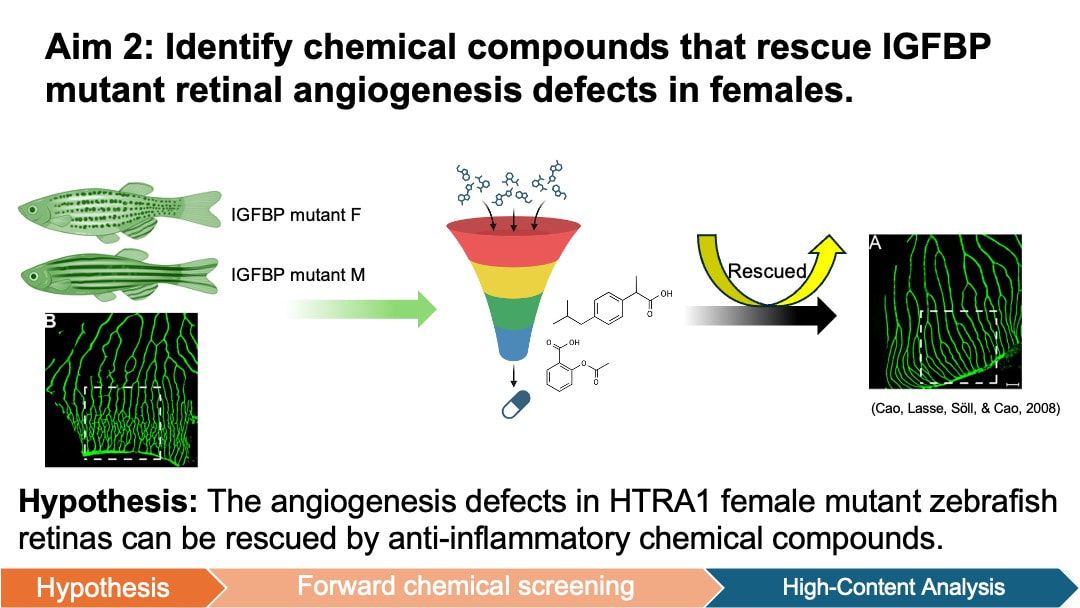
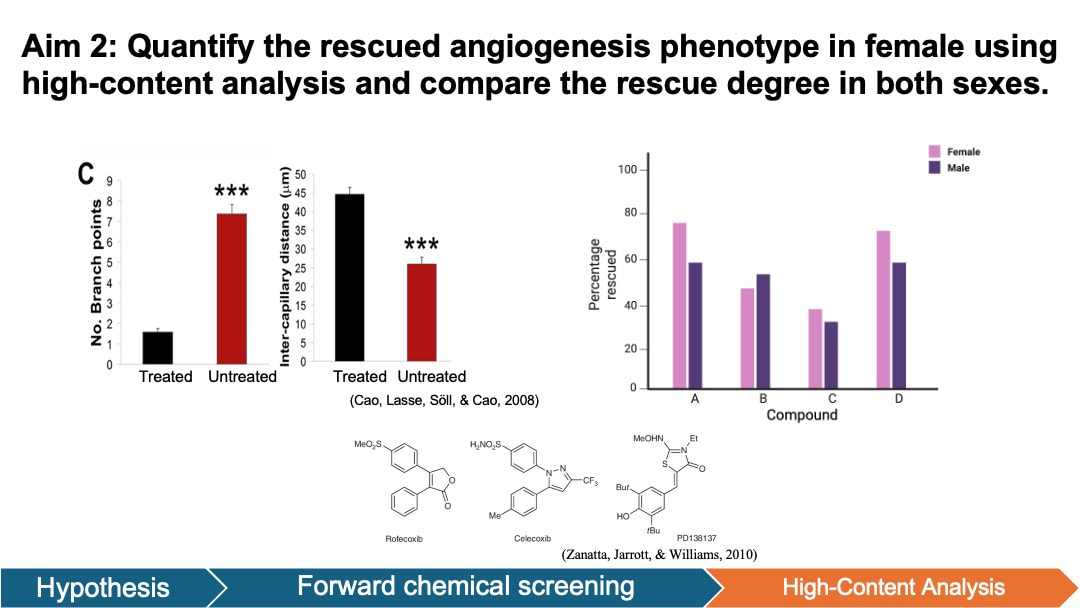
In my aim 2, I hypothesize that the angiogenesis defects in HTRA1 female mutant zebrafish retinas can be rescued by anti-inflammatory chemical compounds. Forward chemical screening is a powerful tool for identifying the molecules that interact with HTRA1 and modulate angiogenesis pathways. The experimentally determined compounds will provide insights into the molecular mechanisms through which HTRA1 influences angiogenesis in zebrafish retinas. Diversity-based libraries are used because it is most used for HTS and has the potential to discover novel chemicals associated with wet AMD. To achieve this aim, I will generate both wild-type (WT) and HTRA1 mutant zebrafish from Aim 1 for both females and males. Then I will conduct a forward small molecule screen to identify compounds from diverse libraries that reduce angiogenesis in the retina of mutant zebrafish.
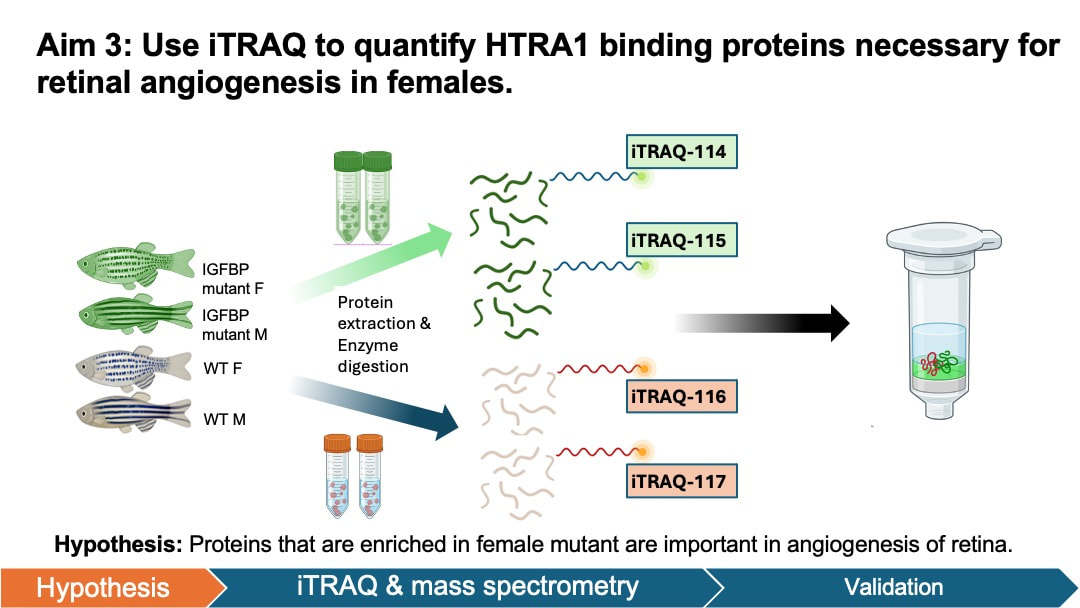
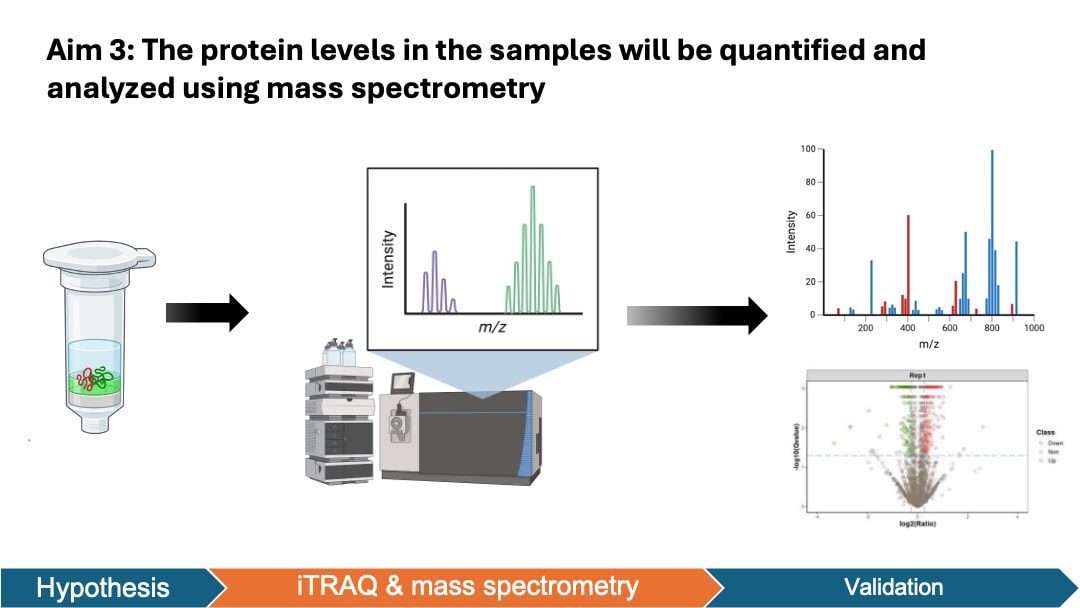
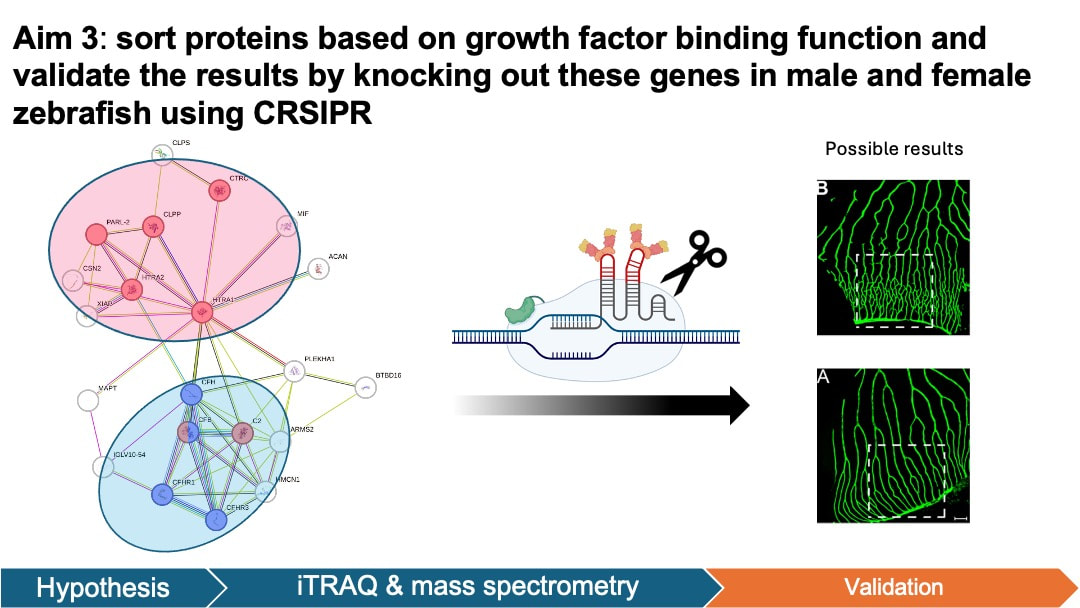
In my aim 3, I hypothesize that proteins that are enriched in female mutant are important in angiogenesis of retina. iTRAQ allows quantification of protein levels in both female and male groups. This will help us identify the key proteins that are elevated in the retinal angiogenesis of female zebrafish. To achieve this aim, I will isolate the retinal pigment epithelium cells tissue from zebrafish groups from aim 1. The protein levels in the samples will be quantified and analyzed using iTRAQ and mass spectrometry. I will then sort proteins based on GO terms and validate the results by knocking out these genes in male and female zebrafish using CRSIPR.
| qiu20042024final.pdf | |
| File Size: | 4713 kb |
| File Type: | |
| qiu20042024final.pptx | |
| File Size: | 6856 kb |
| File Type: | pptx |
| qiu03212024draft2_[autosaved].pdf | |
| File Size: | 6849 kb |
| File Type: | |
| qiu02292024.pptx | |
| File Size: | 2604 kb |
| File Type: | pptx |
Reference:
Header: https://www.psd-dude.com/tutorials/resources/presentation-background.aspx
[1]Fleckenstein, M., Keenan, T.D.L., Guymer, R.H. et al. Age-related macular degeneration. Nat Rev Dis Primers 7, 31 (2021). https://doi.org/10.1038/s41572-021-00265-2
[2] Rudnicka, A. R., Jarrar, Z., Wormald, R., Cook, D. G., Fletcher, A., & Owen, C. G. (2012). Age and Gender Variations in Age-related Macular Degeneration Prevalence in Populations of European Ancestry: A Meta-analysis. Ophthalmology, 119(3), 571–580. doi:10.1016/j.ophtha.2011.09.027
[3] Andrew DeWan et al. ,HTRA1 Promoter Polymorphism in Wet Age-Related Macular Degeneration.Science314,989-992(2006).DOI:10.1126/science.1133807
[4]Rho, J., Percelay, P., Pilkinton, S., Hollingsworth, T. J., Kornblau, I., & Jablonski, M. (2022). An Overview of Age-Related Macular Degeneration: Clinical, Pre-Clinical Animal Models and Bidirectional Translation. IntechOpen EBooks. https://doi.org/10.5772/intechopen.96601
[5]Rastoin, O., Gilles Pagès, & Maeva Dufies. (2020). Experimental Models in Neovascular Age Related Macular Degeneration. International Journal of Molecular Sciences (Online), 21(13), 4627–4627. https://doi.org/10.3390/ijms21134627
[6]Noel, N. C. L., MacDonald, I. M., & W Ted Allison. (2021). Zebrafish Models of Photoreceptor Dysfunction and Degeneration. Biomolecules, 11(1), 78–78. https://doi.org/10.3390/biom11010078
Header: https://www.psd-dude.com/tutorials/resources/presentation-background.aspx
[1]Fleckenstein, M., Keenan, T.D.L., Guymer, R.H. et al. Age-related macular degeneration. Nat Rev Dis Primers 7, 31 (2021). https://doi.org/10.1038/s41572-021-00265-2
[2] Rudnicka, A. R., Jarrar, Z., Wormald, R., Cook, D. G., Fletcher, A., & Owen, C. G. (2012). Age and Gender Variations in Age-related Macular Degeneration Prevalence in Populations of European Ancestry: A Meta-analysis. Ophthalmology, 119(3), 571–580. doi:10.1016/j.ophtha.2011.09.027
[3] Andrew DeWan et al. ,HTRA1 Promoter Polymorphism in Wet Age-Related Macular Degeneration.Science314,989-992(2006).DOI:10.1126/science.1133807
[4]Rho, J., Percelay, P., Pilkinton, S., Hollingsworth, T. J., Kornblau, I., & Jablonski, M. (2022). An Overview of Age-Related Macular Degeneration: Clinical, Pre-Clinical Animal Models and Bidirectional Translation. IntechOpen EBooks. https://doi.org/10.5772/intechopen.96601
[5]Rastoin, O., Gilles Pagès, & Maeva Dufies. (2020). Experimental Models in Neovascular Age Related Macular Degeneration. International Journal of Molecular Sciences (Online), 21(13), 4627–4627. https://doi.org/10.3390/ijms21134627
[6]Noel, N. C. L., MacDonald, I. M., & W Ted Allison. (2021). Zebrafish Models of Photoreceptor Dysfunction and Degeneration. Biomolecules, 11(1), 78–78. https://doi.org/10.3390/biom11010078