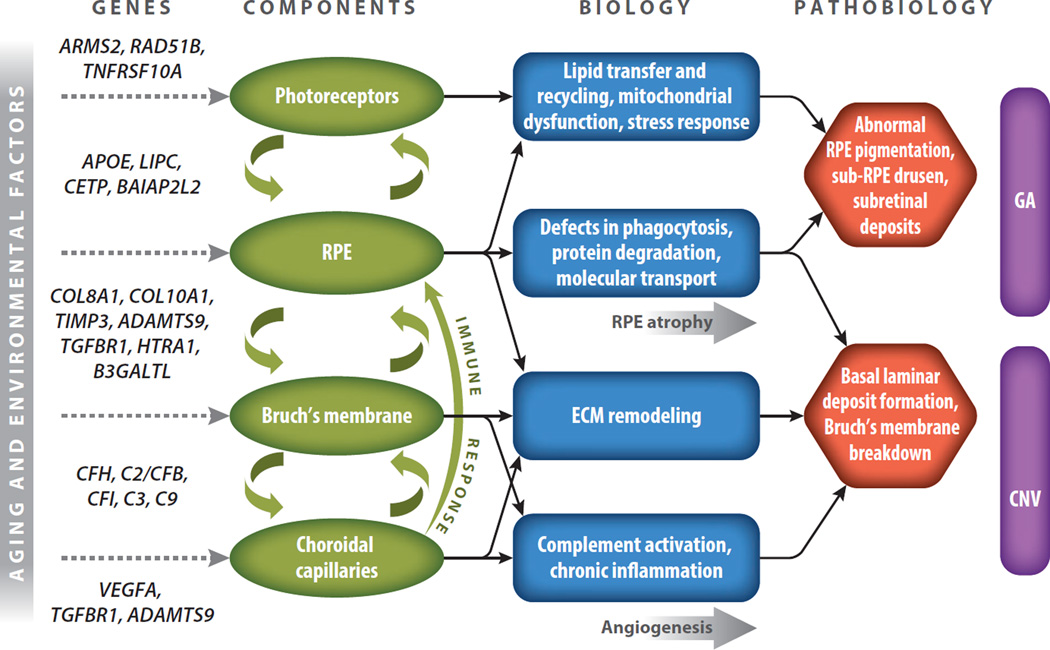
Relevant literature resources regarding AMD & HTRA1
HTRA1 promotes the progression of nAMD through TGF‐β signaling pathway.
One of mechanisms in which HTRA1 contributes to the progression of AMD is through the regulation of TGF-β signaling pathway.
Transforming growth factor-beta (TGFβ) is a secreted cytokine that involves in a variety of physiological processes including. It plays a paradoxical role in carcinogenesisacts: it acts as a tumor suppressor in the early stage of tumorigenesis but becomes tumor-promoting in the later stage. [5] In AMD, TGF-β induces angiogenesis, which is one of major hallmarks of late-stage AMD phenotype. [7] Studies have shown the inhibition of angiogenesis plays a pivotal role in nAMD treatment.[8] The proteomics study identified EGF-containing fibulin-like extracellular matrix 354 protein 1 (EFEMP1) and thrombospondin-1 (THBS1) to be the target of HTRA1 cleavage, which also regulates TGF-β activation. This supports the evidence of HTRA1's regulatory effect on TGF-β.[6]
In bone: HTRA1 regulates bone formation via TGF‐β/BMP signaling, but evidence for both its promoting effect and inhibitory effect in bone formation exist. [1] HTRA1 is also involved in the regulation of cancer as a tumor suppressor associated with TGF‐β signaling pathway. [1]
In brain: Studies have found that HtrA1-dependent proteolysis regulates TGF-β signaling and therefore controls neuronal maturation and developmental survival. [2]. HtrA1 is also highly expressed in the brain during the embryo development.[3]
There is a direct interaction between HtrA1 and TSC2. TSC1-TSC2 complex is critical negative regulator of mTOR signaling pathway. This study identified the intracellular protein TSC2 as a substrate of HtrA1. [4] Although many studies found PDZ domain to be the primary interaction site for HTRA1, this studies found that HtrA1 directly interact with TSC2 through the mac25 domain. [4]
Although elated evidence has shown strong correlation between HTRA1 and TGFb, some studies have reported that HTRA1 does not influence TGF-beta https://link.springer.com/chapter/10.1007/978-3-030-27378-1_1
One of mechanisms in which HTRA1 contributes to the progression of AMD is through the regulation of TGF-β signaling pathway.
Transforming growth factor-beta (TGFβ) is a secreted cytokine that involves in a variety of physiological processes including. It plays a paradoxical role in carcinogenesisacts: it acts as a tumor suppressor in the early stage of tumorigenesis but becomes tumor-promoting in the later stage. [5] In AMD, TGF-β induces angiogenesis, which is one of major hallmarks of late-stage AMD phenotype. [7] Studies have shown the inhibition of angiogenesis plays a pivotal role in nAMD treatment.[8] The proteomics study identified EGF-containing fibulin-like extracellular matrix 354 protein 1 (EFEMP1) and thrombospondin-1 (THBS1) to be the target of HTRA1 cleavage, which also regulates TGF-β activation. This supports the evidence of HTRA1's regulatory effect on TGF-β.[6]
In bone: HTRA1 regulates bone formation via TGF‐β/BMP signaling, but evidence for both its promoting effect and inhibitory effect in bone formation exist. [1] HTRA1 is also involved in the regulation of cancer as a tumor suppressor associated with TGF‐β signaling pathway. [1]
In brain: Studies have found that HtrA1-dependent proteolysis regulates TGF-β signaling and therefore controls neuronal maturation and developmental survival. [2]. HtrA1 is also highly expressed in the brain during the embryo development.[3]
There is a direct interaction between HtrA1 and TSC2. TSC1-TSC2 complex is critical negative regulator of mTOR signaling pathway. This study identified the intracellular protein TSC2 as a substrate of HtrA1. [4] Although many studies found PDZ domain to be the primary interaction site for HTRA1, this studies found that HtrA1 directly interact with TSC2 through the mac25 domain. [4]
Although elated evidence has shown strong correlation between HTRA1 and TGFb, some studies have reported that HTRA1 does not influence TGF-beta https://link.springer.com/chapter/10.1007/978-3-030-27378-1_1
Other review paper on AMD:
Fleckenstein, M., Keenan, T. D. L., Guymer, R. H., Chakravarthy, U., Schmitz-Valckenberg, S., Klaver, C. C., … Chew, E. Y. (2021). Age-related macular degeneration. Nature Reviews Disease Primers, 7(1). doi:10.1038/s41572-021-00265-2
Fleckenstein, M., Keenan, T. D. L., Guymer, R. H., Chakravarthy, U., Schmitz-Valckenberg, S., Klaver, C. C., … Chew, E. Y. (2021). Age-related macular degeneration. Nature Reviews Disease Primers, 7(1). doi:10.1038/s41572-021-00265-2
[1]Li, Y., Yuan, J., Emel Rothzerg, Wu, X., Xu, H., Zhu, S., & Xu, J. (2019). Molecular structure and the role of high‐temperature requirement protein 1 in skeletal disorders and cancers. Cell Proliferation (Print), 53(2). https://doi.org/10.1111/cpr.12746
[2]S. Launay, E. Maubert, N. Lebeurrier, A. Tennstaedt, M. Campioni, F. Docagne, … D. Vivien. (2008). HtrA1-dependent proteolysis of TGF-β controls both neuronal maturation and developmental survival. Cell Death & Differentiation, 15(9), 1408–1416. https://doi.org/10.1038/cdd.2008.82
[3]Campioni, M., Severino, A., Manente, L., Tuduce, I. L., Stefano Toldo, Caraglia, M., … Baldi, A. (2010). The Serine Protease HtrA1 Specifically Interacts and Degrades the Tuberous Sclerosis Complex 2 Protein. Molecular Cancer Research, 8(9), 1248–1260. https://doi.org/10.1158/1541-7786.mcr-09-047
[4]Campioni, M., Severino, A., Manente, L., Tuduce, I. L., Stefano Toldo, Caraglia, M., … Baldi, A. (2010). The Serine Protease HtrA1 Specifically Interacts and Degrades the Tuberous Sclerosis Complex 2 Protein. Molecular Cancer Research, 8(9), 1248–1260. https://doi.org/10.1158/1541-7786.mcr-09-0473
[5]Chaudhury, A., & Howe, P. H. (2009). The tale of transforming growth factor‐beta (TGFβ) signaling: A soigné enigma. IUBMB Life, 61(10), 929–939. https://doi.org/10.1002/iub.239
[6]
May, A., Su, F., Dinh, B., Ehlen, R., Tran, C., Adivikolanu, H., & Shaw, P. X. (2021). Ongoing controversies and recent insights of the ARMS2-HTRA1 locus in age-related macular degeneration. Experimental Eye Research, 210, 108605. doi:10.1016/j.exer.2021.108605
[7]Ferrari, G., Cook, B. D., Vitaly Terushkin, Pintucci, G., & Paolo Mignatti. (2009). Transforming growth factor‐beta 1 (TGF‐β1) induces angiogenesis through vascular endothelial growth factor (VEGF)‐mediated apoptosis. Journal of Cellular Physiology, 219(2), 449–458. https://doi.org/10.1002/jcp.21706
[8]Bressler, S. B. (2009). Introduction: Understanding the Role of Angiogenesis and Antiangiogenic Agents in Age-Related Macular Degeneration. Ophthalmology, 116(10), S1–S7. https://doi.org/10.1016/j.ophtha.2009.06.045
[9]Fritsche, L. G., Fariss, R. N., Stambolian, D., Abecasis, G. R., Curcio, C. A., & Anand Swaroop. (2014). Age-Related Macular Degeneration: Genetics and Biology Coming Together. Annual Review of Genomics and Human Genetics, 15(1), 151–171. https://doi.org/10.1146/annurev-genom-090413-025610
[2]S. Launay, E. Maubert, N. Lebeurrier, A. Tennstaedt, M. Campioni, F. Docagne, … D. Vivien. (2008). HtrA1-dependent proteolysis of TGF-β controls both neuronal maturation and developmental survival. Cell Death & Differentiation, 15(9), 1408–1416. https://doi.org/10.1038/cdd.2008.82
[3]Campioni, M., Severino, A., Manente, L., Tuduce, I. L., Stefano Toldo, Caraglia, M., … Baldi, A. (2010). The Serine Protease HtrA1 Specifically Interacts and Degrades the Tuberous Sclerosis Complex 2 Protein. Molecular Cancer Research, 8(9), 1248–1260. https://doi.org/10.1158/1541-7786.mcr-09-047
[4]Campioni, M., Severino, A., Manente, L., Tuduce, I. L., Stefano Toldo, Caraglia, M., … Baldi, A. (2010). The Serine Protease HtrA1 Specifically Interacts and Degrades the Tuberous Sclerosis Complex 2 Protein. Molecular Cancer Research, 8(9), 1248–1260. https://doi.org/10.1158/1541-7786.mcr-09-0473
[5]Chaudhury, A., & Howe, P. H. (2009). The tale of transforming growth factor‐beta (TGFβ) signaling: A soigné enigma. IUBMB Life, 61(10), 929–939. https://doi.org/10.1002/iub.239
[6]
May, A., Su, F., Dinh, B., Ehlen, R., Tran, C., Adivikolanu, H., & Shaw, P. X. (2021). Ongoing controversies and recent insights of the ARMS2-HTRA1 locus in age-related macular degeneration. Experimental Eye Research, 210, 108605. doi:10.1016/j.exer.2021.108605
[7]Ferrari, G., Cook, B. D., Vitaly Terushkin, Pintucci, G., & Paolo Mignatti. (2009). Transforming growth factor‐beta 1 (TGF‐β1) induces angiogenesis through vascular endothelial growth factor (VEGF)‐mediated apoptosis. Journal of Cellular Physiology, 219(2), 449–458. https://doi.org/10.1002/jcp.21706
[8]Bressler, S. B. (2009). Introduction: Understanding the Role of Angiogenesis and Antiangiogenic Agents in Age-Related Macular Degeneration. Ophthalmology, 116(10), S1–S7. https://doi.org/10.1016/j.ophtha.2009.06.045
[9]Fritsche, L. G., Fariss, R. N., Stambolian, D., Abecasis, G. R., Curcio, C. A., & Anand Swaroop. (2014). Age-Related Macular Degeneration: Genetics and Biology Coming Together. Annual Review of Genomics and Human Genetics, 15(1), 151–171. https://doi.org/10.1146/annurev-genom-090413-025610